Our Research
The Huus Lab will be officially launching at the University of Ottawa, School of Nutrition Sciences in July 2026. Our goal is to combine human cohort studies, big-data approaches and experimental biology to understand how diet-microbiome interactions affect human immune health - from vaccination to chronic disease.
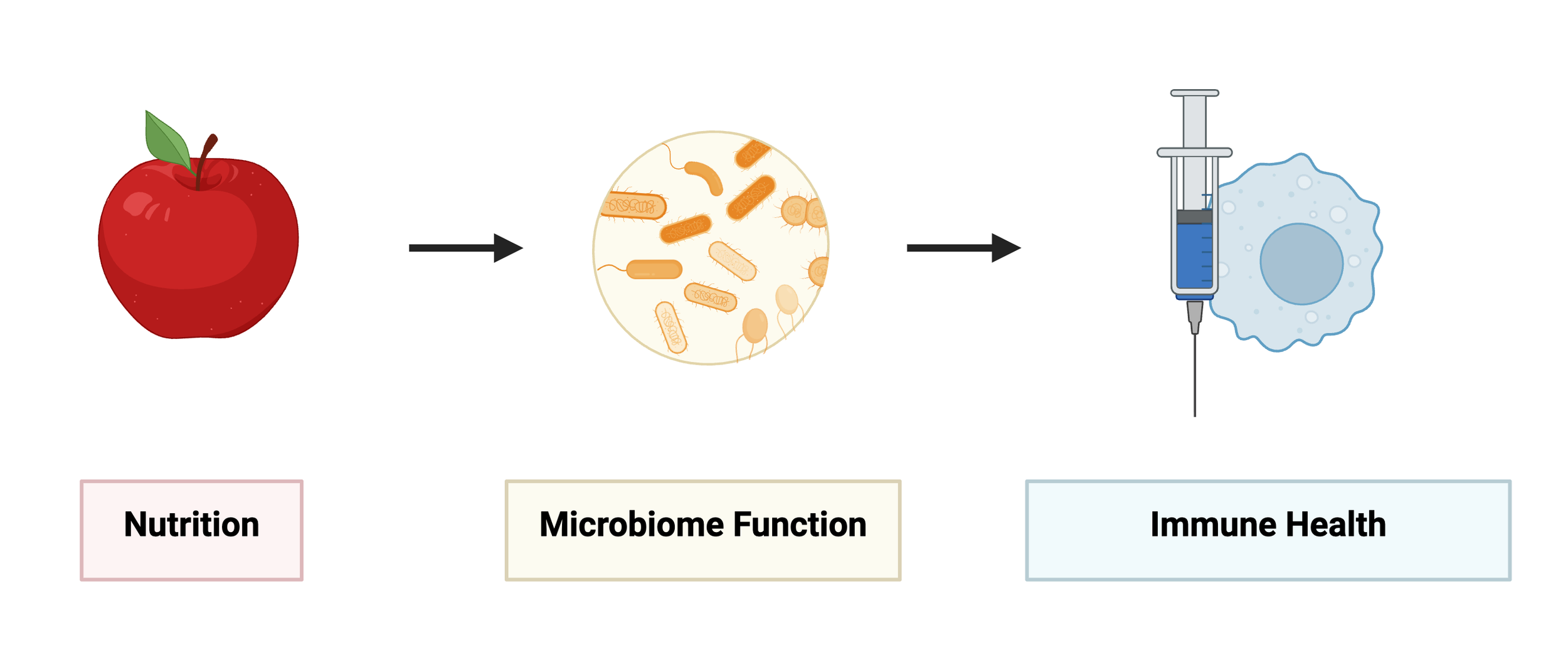
Gut Microbiome and Vaccination
There is enormous variation in how different people respond to the exact same vaccine. This limits vaccine efficacy, leading to insufficient immunity in some recipients and excessive side effects in others. Dr. Huus’ postdoctoral work showed that the risk of a common vaccine side-effect – fever – is influenced by diet-driven activity of the human gut microbiome. In particular, the µHEAT study showed that commensal flagellins stimulate the host innate immune response, increasing the risk of vaccine-induced fever. These flagellins are upregulated in response to industrialized diets, suggesting that dietary interventions might be effective. Going forwards, the Huus Lab aims to understand how these diet-microbiome interactions influence the full spectrum of vaccine responses, including the risk of serious adverse events such as myocarditis, and the downstream efficacy of the adaptive immune system.
Gut Microbiome, Diet, and Chronic Disease

The burden of chronic inflammatory diseases is globally high, and steadily rising. These diseases do not exist in isolation: indeed, inflammatory conditions frequently co-occur. As a member of the Brain-Heart Interconnectome, the Huus lab aims to understand how diet and the gut microbiome affect chronic inflammation and thus susceptibility to chronic diseases of the gut, heart and brain. Preliminary results from the µHEAT cohort (used to study vaccination, above) have revealed chronic inflammatory and cardiometabolic markers associated with the gut microbiome and with dietary habits. Future work in the Huus Lab will attempt to understand how diet and the human microbiome interact to influence the development of chronic inflammatory diseases.

Waltera : a new mystery bug
One key bacterium of interest to the Huus Lab is the genus Waltera. The Waltera genus was only recently described and isolated, but we find it to be strongly associated with vaccine side-effects in human cohorts. In the lab, Waltera produces highly pro-inflammatory flagella in a diet-dependent manner, and experiences a strong growth advantage in the presence of dietary additives present in ultra-processed foods. Together these data suggest that Waltera could be playing an important role in modulating human health and nutrition, but we know very little about this bug. The Huus Lab will take a basic microbiological approach to better characterize Waltera biology, and to further investigate its associations with host health.
Publications
For a full and up-to-date list of publications, please click here
1. Huus KE, µHEAT Clinical Study Group, Tan YH, Abo H, Keller R, Long Vu D, Dauser S, Prokipchuk A, Tyakht A, Youngblut N, Chassaing B, Kang SM, Parsonnet J, Kremsner PG, Maier L, Gewirtz AT, Esen M, Ley RE (2025). The human gut microbiome interacts with diet to prime fever after vaccination. BioRxiv. doi: https://doi.org/10.1101/2025.02.21.639485 (Under Revision, Science)
2. Huus KE and Ley (2024). Gut bacterial metabolism produces neuroactive steroids in pregnant women. Life Metabolism 3(6).
3. Huus KE and Ley RE (2021). Blowing hot and cold: Body temperature and the microbiome. mSystems. e00707-21.
4. Huus KE*, Hoang TT*, Knuff K, Vogt S, Cirstea M, Sansonetti PJ, Vonaesch P, Finlay BB (2021). Cross-feeding between intestinal pathobionts promotes their overgrowth during undernutrition. Nat Comm 12:6860. (*authors contributed equally).
5. Huus KE, Petersen C, Finlay BB (2021). Diversity and dynamism of IgA-microbiota interactions. Nat Rev Immunol. doi: 10.1038/s41577-021-00506-1.
6. Huus KE, Frankowski M, Pucuc-Bakovic M, Vuckovic F, Lauc G, Mullish B, Monaghan TM, Kao D, Finlay BB (2021). Changes in IgA-targeted microbiota following fecal transplantation for recurrent Clostridioides difficile infection. Gut Microbes. 13(1):1-12.
7. Huus KE, Rodriguez-Pozo A, Kapel N, Nestoret A, Habib A, Dede M, Manges A, Collard JM, Sansonetti PJ, Vonaesch P, Finlay BB (2020). Immunoglobulin recognition of fecal bacteria from stunted and non-stunted children: findings from the Afribiota study. Microbiome. 8:113. doi: 10.1186/s40168-020-00890-1.
8. Huus KE, Bauer KC, Brown EM, Bozorghmehr T, Woodward S, Serapio A, Boutin RCT, Petersen C, Finlay BB (2020). Commensal bacteria modulate immunoglobulin A binding in response to host nutrition. Cell Host & Microbe. doi: 10.1016/j.chom.2020.03.012.
9. Huus KE and Finlay BB (2017). Feeding the microbial multitudes: co-infection in a malnourished host. Nat Rev Gastroenterol Hepatol. doi: 10.1038/nrgastro.2017.133.
10. Huus KE, Joseph J, Zhang L, Wong A, Aaron SD, Mah TF, Sad S. (2016). Clinical isolates of Pseudomonas aeruginosa from chronically infected cystic fibrosis patients fail to activate the inflammasome during both stable infection and pulmonary exacerbation. Journal of Immunology. doi: 10.4049/jimmunol. 1501642.